
Some isotopes are unstable and will lose protons, other subatomic particles, or energy to form more stable elements. These two alternate forms of carbon are isotopes. Therefore, it has a mass number of 14 (six protons and eight neutrons) and an atomic number of 6, meaning it is still the element carbon. Carbon-14 contains six protons and eight neutrons. Therefore, it has a mass number of 12 (six protons and six neutrons) and an atomic number of 6 (which makes it carbon). Carbon-12, the most common isotope of carbon, contains six protons and six neutrons. Some elements, such as carbon, potassium, and uranium, have naturally occurring isotopes. Isotopes are different forms of the same element that have the same number of protons, but a different number of neutrons. The arrangement of the table also shows how the electrons in each element are organized and provides important details about how atoms will react with each other to form molecules. The periodic table also provides key information about the properties of elements -often indicated by color-coding. Because of these characteristics, the elements are arranged into the periodic table of elements, a chart of the elements that includes the atomic number and relative atomic mass of each element. How they combine is based on the number of electrons present. Some form specific types of bonds, whereas others do not. Different elements have different melting and boiling points, and are in different states (liquid, solid, or gas) at room temperature. These numbers provide information about the elements and how they will react when combined. Therefore, it is possible to determine the number of neutrons by subtracting the atomic number from the mass number. The mass number, or atomic mass, is the number of protons plus the number of neutrons of that element. 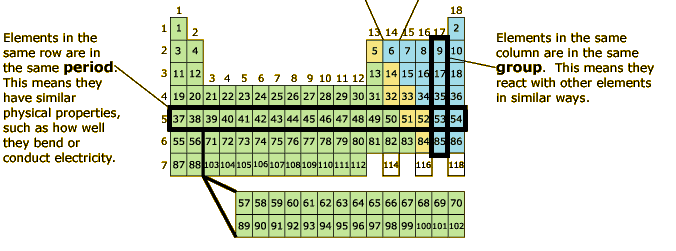
The atomic number of an element is equal to the number of protons that element contains. Each contains a different number of protons and neutrons, giving it its own atomic number and mass number. The number of electrons does not factor into the overall mass, because their mass is so small.Īs stated earlier, each element has its own unique properties. The positive (protons) and negative (electrons) charges balance each other in a neutral atom, which has a net zero charge.īecause protons and neutrons each have a mass of 1, the mass of an atom is equal to the number of protons and neutrons of that atom. Neutrons, like protons, reside in the nucleus of an atom. Figure 2.2 Atoms are made up of protons and neutrons located within the nucleus, and electrons surrounding the nucleus. It has a negligible mass and has a charge of –1. In other words, it resides outside of the nucleus. An electron is a negatively charged particle that travels in the space around the nucleus. A proton is a positively charged particle that resides in the nucleus (the core of the atom) of an atom and has a mass of 1 and a charge of +1. The only exception is hydrogen (H), which is made of one proton and one electron. These combinations continue until entire multicellular organisms are formed.Īll atoms contain protons, electrons, and neutrons. In multicellular organisms, such as animals, molecules can interact to form cells that combine to form tissues, which make up organs. They contain atoms that combine together to form molecules. If a hydrogen atom were broken down into subatomic particles, it would no longer have the properties of hydrogen.Īt the most basic level, all organisms are made of a combination of elements. Hydrogen atoms cannot be broken down into anything smaller while still retaining the properties of hydrogen. For example, one hydrogen atom has all of the properties of the element hydrogen, such as it exists as a gas at room temperature, and it bonds with oxygen to create a water molecule. 
AtomsĪn atom is the smallest component of an element that retains all of the chemical properties of that element. 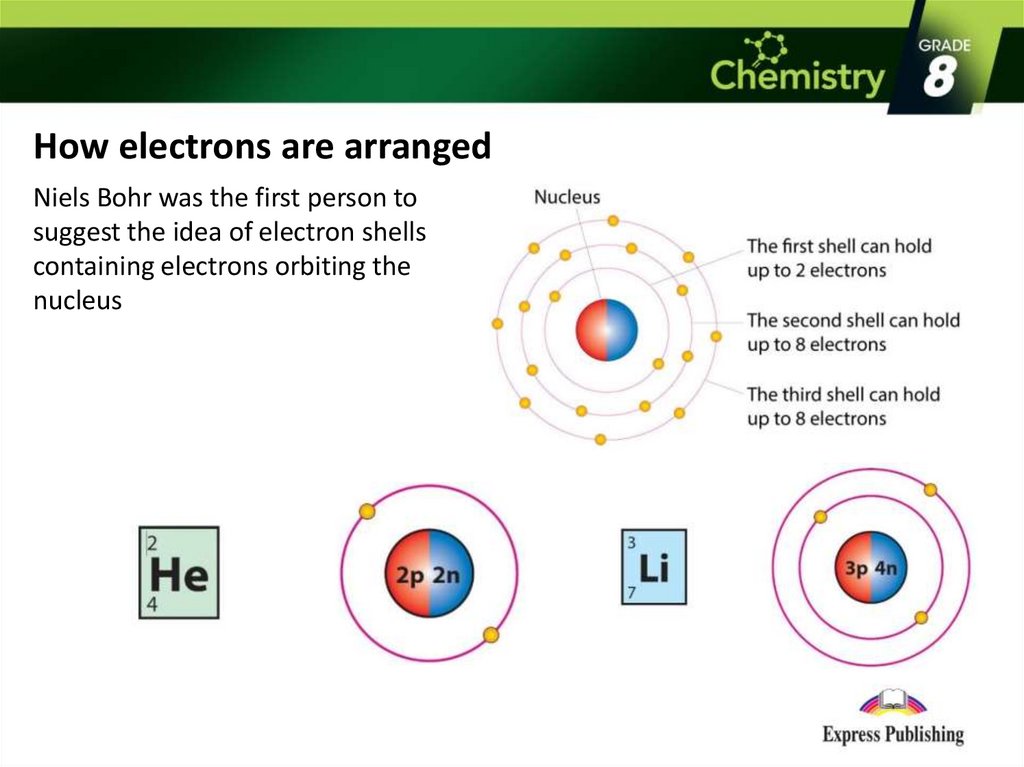
These unique properties allow elements to combine and to bond with each other in specific ways. The remaining 26 elements are unstable and, therefore, do not exist for very long or are theoretical and have yet to be detected.Įach element is designated by its chemical symbol (such as H, N, O, C, and Na), and possesses unique properties. A total of 118 elements have been defined however, only 92 occur naturally, and fewer than 30 are found in living cells. Each element is made of atoms, each with a constant number of protons and unique properties. All matter is composed of elements, substances that cannot be broken down or transformed chemically into other substances. At its most fundamental level, life is made up of matter.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
